
Goutam Narayan Tumulu
Prime Minister Research Fellow at IIT Bombay

©Goutam Narayan
About Me
I work on characterization and performance evaluation of Solid-acid catalysts to understand structure-activity correlations for design of optimal catalysts.
Research Interests

By Myasein at en.wikipedia - Transferred from en.wikipedia to Commons by Ronhjones., CC BY-SA 3.0, https://commons.wikimedia.org/w/index.php?curid=17420804
The acid strength of the catalyst must correspond precisely with the basic strength of the reactant for maximum conversion and selectivity. Many reactions require strong acid sites to proceed. This has made Tungsten Oxides and Heteropolyacids popular, since they possess superacidity (Acidity>100% H2SO4). We will be interested in tuning the acidity of Tungsten based acids by immobilising on various supports and conjugating with different metals. We intend to employ both theoretical(DFT) and physical experiments for the screening and validating optimum Metal-Acid-Support combinations for a reaction.
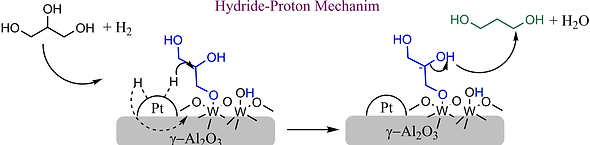
Catalysts 2021, 11(10), 1171; https://doi.org/10.3390/catal11101171
Solid acids are acids which can liberate H+ ions in the reaction medium without any significant change to their structure, preserving their heterogeneity. They can catalyse almost all reactions catalysed by liquid acids. The catalytic activity combined with heterogeneous nature makes solid acids a great choice for commercial applications. Metal nanoparticles can be supported on these solid acids to make them bifunctional and catalyse complex reactions which require hydrogenation/oxidation steps.Our lab group intends to search and optimise Metal-Solid Acid bifunctional catalysts for important reactions.
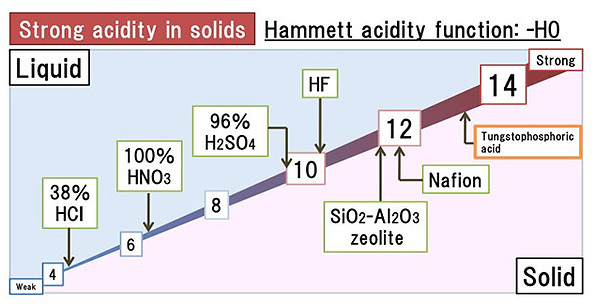
(JAPAN NEW METALS CO.,LTD,http://www.jnm.co.jp/en/products/heteropoly_acids)
Courses Taken
Cumulative CPI=8.84

Publications

Abstract
The 31P NMR Chemical Shifts(δP) of Trimethylphosphine Oxide (TMPO) conjugated with ten common liquid molecular acids have been calculated employing the Density Functional Theory (DFT) based computational method. These shifts have been correlated with the intrinsic acid strength parameter, Deprotonation Energy (DPE), of the underlying acids and the complexation energy of TMPO (ΔΕ). The acids, anions, and conjugated complexes were modeled with PW91 functional and 6-31++G(d,p) basis set. The NMR calculations were performed on the relaxed structures as single-point calculations using the Gauge Including Atomic Orbital (GIAO) method at the MP2 level and TZVP basis set. Poor linearity was observed in the correlation of δP with respect to DPE (R2=0.867), while strong linearity was seen with the complexation energy ΔΕ (R2=0.967). We have hypothesized the unreliable modeling of anions and Non-Covalent Interactions (NCI) to be the factors affecting the linear trend of δP with respect to DPE. To support our hypothesis, we have presented Reduced Density Gradient (RDG) Maps and 31P NMR Tensor spatial orientation data of the Acid-TMPO conjugates.
Narayan Tumulu, G. et al. Role of Non-Covalent Interactions on Acid Strength Measurement of Liquid Acids using Phosphine Oxide NMR Probe. ChemistrySelect 9, e202304804 (2024).
https://doi.org/10.1002/slct.202304804
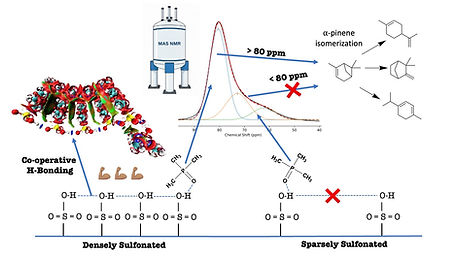
Tumulu, G. N. et al. Effect of sulfonation density on acid strength in ion exchange resins: Insights from solid-state NMR and density functional theory. Molecular Catalysis 593, 115794 (2026).
https://doi.org/10.1016/j.mcat.2026.115794
Abstract
Teaching Duties
Guest Faculty at TKIET Warananagar(Affl. to Shivaji University)
Course: Chemical Reaction Engineering I
Credits: 4
Total: 36 hrs
Guest Faculty at TKIET Warananagar(Affl. to Shivaji University)
Course: Transport Phenomenon
Credits: 4
Total: 48 hrs
PMRF TA at Science For Rural India Programme (SFRI)
Topic: First Law of Thermodynamics & Perpetual Machines
Total: 48 hrs
Guest Speaker at SAVITRIBAI PHULE PUNE UNIVERSITY
Topic: Nanotechnology:Fundamentals of Nanotechnology and its Legal Aspects
Total: 2 hrs
Guest Faculty at MIT World Peace University, Pune
Course: Research Methodology For Chemical Engineers
Total: 96 hrs
Guest Faculty at Thadomal Shahani Engineering College
Course: Chemical Reaction Engineering II
Total: 15 hrs
Guest Faculty at Thadomal Shahani Engineering College
Course: Heat & Pressure Effects in Reactor Design
Total: 11 hrs
